New method offers a promising alternative for upgrading natural gas
已发布 16 六月, 2022
Methane gas is considered a clean energy due to its high hydrogen-to-carbon ratio and low carbon dioxide (CO2) emissions. This has seen the appetite for methane grow in recent years, as countries strive to meet ambitious climate change targets.
Natural gas is a critical source of methane; it consists primarily of methane with just a small volume of ethane (C2) and propane (C3) gas impurities. To meet industry standards, these impurities must be removed prior to the transportation and use of methane. Capturing them effectively is important, not only for the environment, but because these hydrocarbon gases yield vital raw materials for other processes.
Due to differences in the various gases’ boiling points, the removal of C2 and C3 is typically performed using cryogenic distillation. However, that cryogenic process is energy- and cost-intensive. In fact, distillation-based separation procedures currently account for 10–15% of global energy consumption. As a result, the hunt is on for cheaper and more environmentally friendly separation methods.
In a study published in the KeAi journal Green Chemical Engineering, researchers describe an innovative, copper-based metal organic framework (Cu-MOF) they have developed to separate propane and ethane from methane. The MOF exhibits a high adsorption capacity and selectivity for C2–C3 hydrocarbons. And the adsorption isotherms at ambient conditions show a remarkable uptake (C3H8 of 134.0 cm3 g-1) and excellent selectivity of 204 and nine for C3H8/CH4 and C2H6/CH4.
Corresponding author Qing-Yuan Yang, from Xi’an Jiaotong University in China, explains: “Our Cu-MOF showed a significant difference in adsorption capacity for light hydrocarbons, while displaying remarkable selectivity for C3H8 and C2H6 over CH4. More importantly, cycling breakthrough experiments have demonstrated that the Cu-MOF can efficiently separate ethane and propane from mixtures of C1/C2/C3 with optimal cycling performance.”
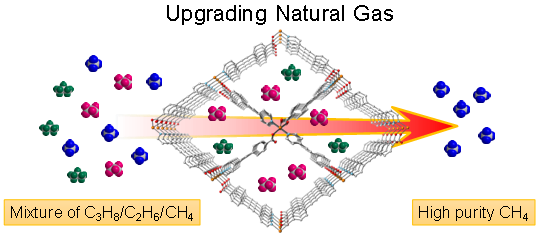
Image: Researchers have developed an energy- and cost-effective process to create methane from natural gas. Credit: Xi’an Jiaotong University, China.
###
Contact the corresponding author: Qing-Yuan Yang, qingyuan.yang@xjtu.edu.cn