Lenalidomide promotes melarsoprol-activated cGAS-STING-mediated immunotherapy for hepatocellular carcinoma
Published 31 October, 2025
Hepatocellular carcinoma (HCC), the most common primary liver cancer, poses a global health threat with over 680,000 new diagnoses and 620,000 deaths annually. Current immunotherapies often fail due to the liver's inherent immune tolerance and the immunosuppressive tumor microenvironment (TME)—a barrier that suppresses anti-tumor immune responses.
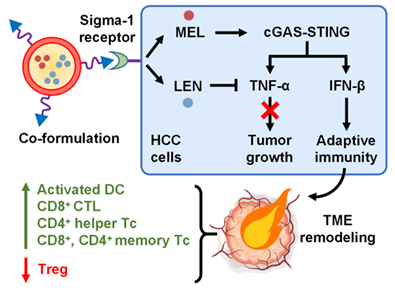
The cGAS-STING pathway has been identified as a critical target to “unlock” the immunosuppressive TME. That is, when cGAS detects damaged DNA in tumor cells, it triggers STING-mediated production of type I interferons (e.g., IFN-β), which in turn activates dendritic cells (DCs) and cytotoxic T lymphocytes (CTLs) to attack cancer cells. Nonetheless, while some chemother drugs can activate this pathway, few have been validated for HCC.
To that end, a research team led by Dr. Zhuo Yu from Shuguang Hospital, Shanghai University of Traditional Chinese Medicine, and Prof. Jianfeng Guo from Jilin University, developed a novel nanoparticle system to co-deliver two drugs: melarsoprol (MEL), which activates cGAS-STING, and lenalidomide (LEN), which suppresses TNF-α.
The study was published in Fundamental Research.
“We found that while MEL activates the immune system, it also overproduces TNF-α, which can fuel the tumor,” explains Yu. “By adding LEN, we were able to block this harmful effect while preserving the anticancer immune response.”
The team encapsulated both drugs into red blood cell membrane-coated nanoparticles, which targeted the tumor and released the drugs in response to the acidic tumor environment.
“In mouse models of liver cancer, the combination therapy significantly reduced tumor growth, increased immune cell infiltration, and improved survival—without causing noticeable toxicity,” says Yu.
“This nanoparticle system allows us to deliver both drugs precisely to the tumor, maximizing their effect while minimizing side effects,” adds Guo. “It's a step toward more effective and safer combination therapies for HCC.”
The team plans to advance the co-formulation to clinical trials, with a focus on combining it with existing immunotherapies (e.g., immune checkpoint inhibitors) for even greater efficacy.
Contact author: Jianfeng Guo, School of Pharmaceutical Sciences, Jilin University, Changchun 130021, China, jguo@jlu.edu.cn
Funder: This work was supported by National Natural Science Foundation of China (82222074, 82074154 and 81774240), the Siming Scholar from Shanghai Shuguang Hospital (SGXZ-201904), the Youth Tip-top Talent Program from Shanghai Municipal Commission of Health, the Constant-eminent Program from Shanghai Municipal Commission of Health, the Xinglin Youth Scholar from Shanghai University of Traditional Chinese Medicine, and the Norman Bethune Program of Jilin University, China (2022B40).
Conflict of interest: The authors declare that they have no conflicts of interest in this work.
See the article: Yu Z, Zou Y F, Han S L, et al. Lenalidomide promotes melarsoprol-activated cGAS-STING-mediated immunotherapy for hepatocellular carcinoma via attenuating TNF-α activity [J]. Fundamental Research, Volume 5, Issue 3, March 2025, Pages 1298-1312, https://doi.org/10.1016/j.fmre.2023.05.013

