Pulling a hasty escape: The role of a novel long non-coding RNA in the immune escape of pathogenic Vibrio in fish
Published 17 January, 2023
MicroRNAs (miRNA) are small non-coding RNAs that regulate almost all biological processes, protein production, inflammatory responses, immune regulation, tumorigenesis and infection. In mammals, the classic formation of miRNA needs to transcribe a long primary miRNA in the nucleus and then process it into hairpin RNA with about 60–70 nucleotides. Eventually, this precursor miRNA will be transported to the cytoplasm for processing and shearing resulting in the generation of mature miRNA.
In recent decades, there have been reports of an alternative way of generating miRNA. The coding genes of some miRNAs are actually located in the coding genes of long non-coding RNA (lncRNA) and transcribed together with lncRNA. There is also increasing evidence of lncRNA playing a critical and irreplaceable role in innate immunity in mammals. Such evidence in lower vertebrates, such as the teleost fish (ray-finned fish) is, however, lacking.
To that end, a research team from Shanghai Ocean University Tled by Tianjun Xu, set out to elucidate the functions and characteristics of IncRNa in the teleost fish.
“We have identified a lncRNA-miRNA-mRNA axis regulatory network involved in antibacterial responses in teleost fish miiuy croaker, an important species of marine fish that supports capture fisheries and aquaculture,” shares Xu. “A novel immune-related lncRNA MIR144HG was shown to negatively regulate the antibacterial immunity of miiuy croaker caused by the bacteria Vibrio anguillarum and Vibrio harveyi—the two most susceptible Gram-negative pathogens in aquaculture. The economic losses caused by these two bacteria every year are immeasurable.”
Previous reports of lncRNA in teleost fish shown that it mostly positively regulates the innate immune response. This is the first time that a non-coding RNA has been found to promote the escape of the two bacteria in a teleost. Furthermore, the team demonstrated that MIR144HG can function as a precursor of miR-144–3p to produce miR-144–3p and enhance the inhibitory effect of miR-144–3p on the proteins MyD88, TAK1 and p65, and thus inhibit the antibacterial immune response.
There was also evidence of MyD88 and p65 participating in the regulation of innate immune response caused by the two Vibrio bacteria, with both proteins having an important role in combatting invasion by V. anguillarum.
The team’s findings are published in the KeAi journal Water Biology and Security.
“This study not only clarifies the mechanisms of the lncRNA-miRNA-mRNA axis in antibacterial immune responses, but also sheds light on the impact of lncRNA on host immunity and bacterial escape,” says Xu.
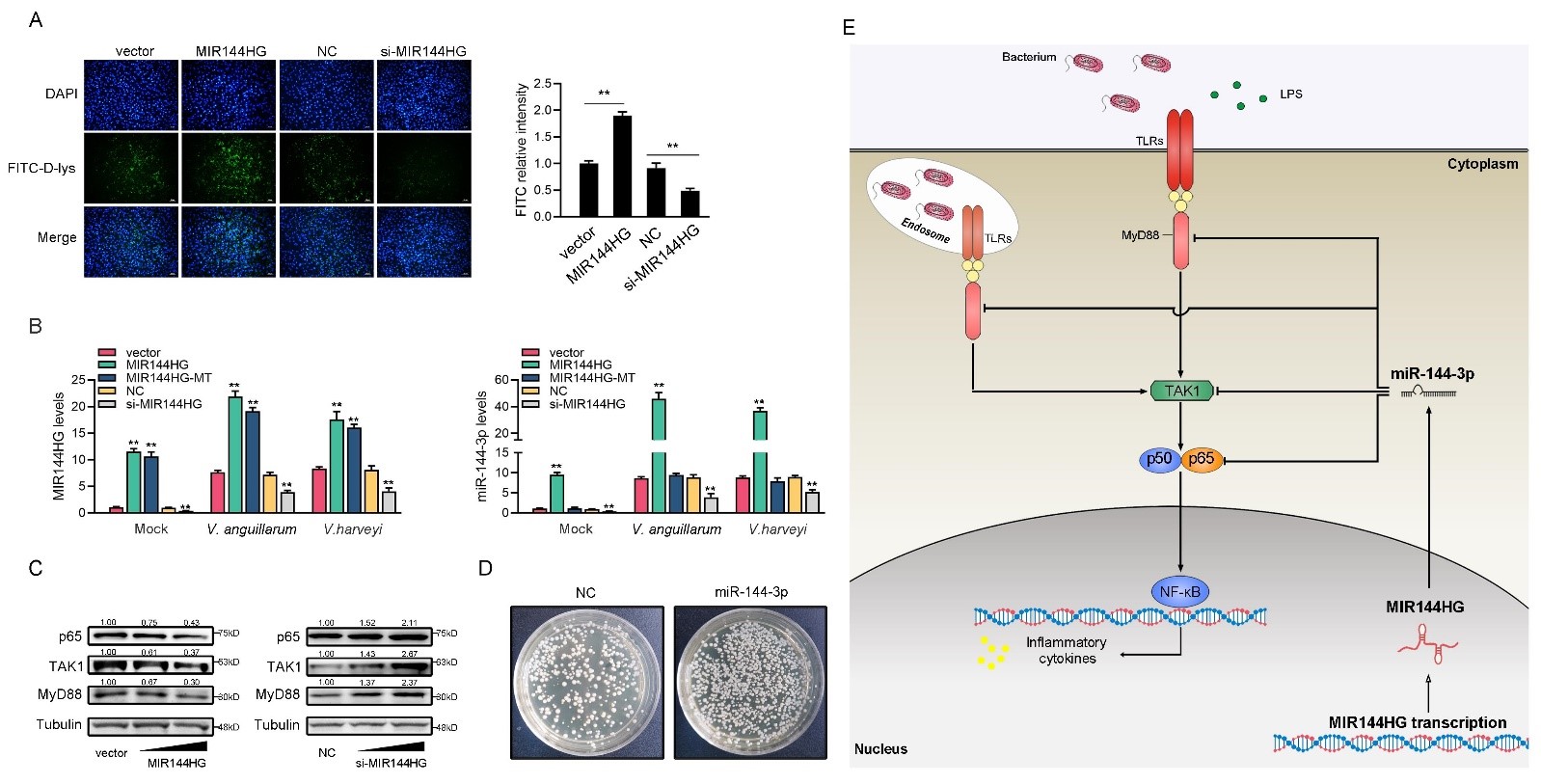
Image: LncRNA MIR144HG regulates the antibacterial innate immune mediated by MyD88, TAK1, and p65 by deriving miR-144-3p in fish
Contact author name, affiliation, email address: Tianjun Xu, Laboratory of Fish Molecular Immunology, College of Fisheries and Life Science, Shanghai Ocean University, Shanghai, China, tianjunxu@163.com.